Biology Unveiled
Poster Session A
(1018-A) A HCS platform for systematic analysis of 3D spheroids at subcellular resolution
Wednesday, May 24, 2023
13:30 - 14:30 CET
Location: Hall 3
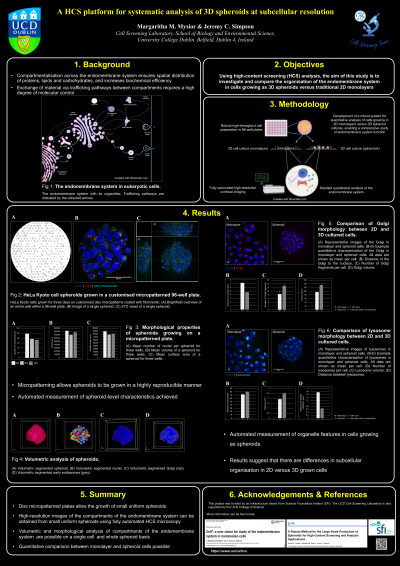
Abstract: The human body is a complex assembly of tissues and organs, each of which is made of different types of cells. Specialisation of function is achieved via compartmentalisation within each cell, as well as defined communication networks that exist between endomembrane compartments. The endomembrane system is essential for synthesising and distributing proteins, lipids, and carbohydrates, and as such, it needs to be tightly regulated. It is, therefore essential to understand the details of how internal compartments achieve homeostasis if we are to ultimately decipher the molecular basis of the large number of human diseases associated with dysfunction of the endomembrane system. To date, much of our understanding of this network of membranes has come from cells growing as traditional two-dimensional (2D) monolayers; however, this scenario does not represent the arrangement of cells growing in tissues in our body. We have developed a robust system for preparation and automated high-resolution quantitative fluorescence imaging of small three-dimensional (3D) cell culture models (spheroids), which can be used in a variety of studies (1). We demonstrate that HeLa Kyoto cells growing on micropatterned plates coated with fibronectin form spheroids of a consistent size and shape, thereby making them highly suitable for automated imaging and screening. We further show that we can use high-content screening microscopy to accurately quantify the spatial distribution and morphological features of a range of organelles in each cell within the spheroids. We compare these results with those from cells grown as traditional 2D monolayers, to examine what changes occur at the subcellular level when cells are grown in this different environment. Additionally, we show that we can quantify changes to the endomembrane system under perturbation conditions (e.g., using RNA interference and drug treatments). We believe that the platform that we have developed could have wide-ranging applications, not only in furthering our fundamental understanding of cell function but also in assessing disease and therapeutic efficacy in a multicellular 3D context.
(1) Mysior MM & Simpson JC (2021). Cell3: A new vision for study of the endomembrane system in mammalian cells. Bioscience Reports 41(12):BSR20210850C.
(1) Mysior MM & Simpson JC (2021). Cell3: A new vision for study of the endomembrane system in mammalian cells. Bioscience Reports 41(12):BSR20210850C.
- MM
Margaritha Mysior, N/A (she/her/hers)
PhD
University College Dublin
Dublin, Dublin, Ireland
